PENDING REVISION 2024
Introduction
The goal of feminizing hormone therapy is the development of female secondary sex characteristics, and suppression/minimization of male secondary sex characteristics. General effects include breast development (usually to Tanner stage 2 or 3), a redistribution of facial and body subcutaneous fat, reduction of muscle mass, reduction of body hair (and to a lesser extent, facial hair), change in sweat and odor patterns, and arrest and possible reversal of scalp hair loss. Sexual and gonadal effects include reduction in erectile function, changes in libido, reduced or absent sperm count and ejaculatory fluid, and reduced testicular size. Feminizing hormone therapy also brings about changes in emotional and social functioning. The general approach of therapy is to combine an estrogen with an androgen blocker, and in some cases a progestogen.
Estrogens
The primary class of estrogen used for feminizing therapy is 17-beta estradiol, which is a "bioidentical" hormone in that it is chemically identical to that from a human ovary. The general approach is similar to estrogen replacement in agonadal (i.e., Turner syndrome) or menopausal states, with some dosing modifications. 17-beta estradiol (or simply estradiol) is most commonly delivered to transgender women via a transdermal patch, oral tablet, or injection of a conjugated ester (estradiol valerate or estradiol cypionate). No outcome studies have been conducted on injectable estradiol valerate or cypionate, presumably due to their uncommon modern use outside of transgender care settings; due to this limited use, manufacturers have little incentive to produce this medicine, and shortages have been reported. Other delivery routes for estradiol such as transdermal gel or spray are formulated for the treatment of menopausal vasomotor symptoms and while convenient and effective in some transgender women, in others these routes may not be able to achieve blood levels in the physiologic female range. Compounded topical creams and gels also exist from specialty pharmacies; if these are to be used it is recommended that the prescriber consult with the compounding pharmacist to understand the specific details and dosing of the individual preparation. Compounded estradiol valerate or cypionate for injection also exists, and may be an alternative in times of shortage or more cost effective for those who must pay cash for their prescriptions.
Conjugated equine estrogens (Premarin®) have been used in the past but are not recommended for a number of reasons, including inability to accurately measure blood levels and some suggestion of increased thrombogenicity and cardiovascular risk.[1,2] Equine estrogens are obtained from the urine of pregnant, catheterized horses; no evidence exists to suggest that these estrogens are superior to bioidentical human estradiol. Ethical concerns have been raised regarding the methods of production of equine estrogens. Ethinyl estradiol is a synthetic estrogen used in contraceptive preparations and is associated with an increased thrombotic risk.[3,4] In the context of contraception, ethinyl estradiol has more consistent and reliable cycle control and as such is better tolerated, balancing out the potentially increased risk of VTE.[5] In the setting of gender affirmation there is no need for cycle or bleeding regulation, and thus the use of ethinyl estradiol and its inherent risks are not warranted (Grading: T O S).
Side effects of estrogens may include migraines, mood swings, hot flashes, and weight gain.
Antiandrogens - common approaches
Suppression of testosterone production and blocking of its effects contributes to the suppression / minimization of male secondary sexual characteristics. Unfortunately, many of these characteristics are permanent upon completion of natal puberty and are irreversible. Androgen blockers allow the use of lower estradiol dosing, in contrast to the supraphysiologic estrogen levels (and associated risks) previously used to affect pituitary gonadotropin suppression.[6]
Spironolactone is the most commonly used androgen blocker in the U.S. Spironolactone is a potassium sparing diuretic, which in higher doses also has direct anti-androgen receptor activity as well as a suppressive effect on testosterone synthesis.[6] Doses of 200mg daily in non-transgender women being treated for hair loss have been described as safe, though doses of up to 400mg/day have been reported without negative effect.[7] Hyperkalemia is the most serious risk but is very uncommon when precaution is taken to avoid use in individuals with renal insufficiency, and use with caution and frequent monitoring in those on ACE inhibitor or ARB type medications. Due to its diuretic effect, patients may experience self-limited polyuria, polydipsia, or orthostasis.
5-alpha reductase inhibitors include finasteride and dutasteride. Finasteride blocks 5-alpha reductase type 2 and 3 mediated conversion of testosterone to the potent androgen dihydrotestosterone.[8] Finasteride 1mg daily is FDA-approved for male pattern baldness, while the 5mg dose is approved for management of prostatic hypertrophy.[9] Dutasteride 0.5mg more effectively blocks the type 1 isozyme, which is present in the pilosebaceous unit and therefore may have more dramatic feminizing effects. Since these medications block neither the production nor action of testosterone, their antiandrogen effect is less than that encountered with full blockade. 5-alpha reductase inhibitors may be a good choice for those unable to tolerate, or with contraindications to the use of spironolactone. 5-alpha reductase inhibitors may also be an option for use as a single agent in patients seeking partial feminization, or for those who continue to exhibit virilized features or hair loss after complete androgen blockade or orchiectomy.
Antiandrogens - other approaches
Antiandrogens can also be used alone to bring reduced masculinization and minimal breast development, or in those patients who wish to first explore reduced testosterone levels alone, or in those with contraindications to estrogen therapy. In the absence of estrogen replacement, some patients may have unpleasant symptoms of hot flashes and low mood or energy. Long term full androgen blockade without hormone replacement in men who have undergone treatment for prostate cancer results in bone loss, and this effect would also be expected to occur in transgender individuals.[10] In addition to titrating dosing to both clinical effect and testosterone levels as guided by patient goals, monitoring hormone levels to insure suppressed gonadotropins (luteinizing hormone [LH] and follicle stimulating hormone [FSH]) levels may serve as a surrogate marker to indicate adequate sex hormone levels for maintaining bone density in such patients (Grading: T O W).[11]
In many countries, cyproterone acetate, a synthetic progestogen with strong anti-androgen activity is commonly used. Cyproterone has been associated with uncommon episodes of fulminant hepatitis.[12] Bicalutamide, a direct anti-androgen used for the treatment of prostate cancer, also has a small but not fully quantified risk of liver function abnormalities (including several cases of fulminant hepatitis); while such risks are acceptable when considering the benefits of bicalutamide in the management of prostate cancer, such risks are less justified in the context of gender affirming treatment.[13] No evidence at present exists to inform such an analysis.
In some patients, complete androgen blockade may be difficult or even impossible using standard regimens. In cases of persistent elevations of testosterone in the setting of maximal antiandrogen dosing with good medication adherence, autonomous endogenous production (i.e. tumor) as well as undisclosed exogenous testosterone (i.e. to maintain erectile function) should be considered. An evaluation for testicular neoplasms should be performed with a scrotal exam as well as testing for elevated serum human choriogonadotropin (hCG), lactate dehydrogenase (LDH), alpha-fetoprotein (AFP) levels, and possibly scrotal imaging.[14] Once these causes have been ruled out, additional options can include gonadotropin releasing hormone analogues (GnRH) or orchiectomy. GnRH analogs are used routinely in the care of peripubertal transgender youth who require pubertal delay,[15] and have been described in the care of transgender adults as well.[16] Drawbacks to the use of GnRH analogs is primarily related to cost and difficulties in obtaining insurance coverage, as well as the need for either repeated injections, multiple daily nasal sprays, or surgical implantation. Orchiectomy may represent an ideal option in transgender women who do not desire to retain their gonads; this brief, inexpensive, outpatient procedure requires only several days for recovery and does not preclude future vaginoplasty.
Progestogens: There have been no well-designed studies of the role of progestogens in feminizing hormone regimens. Many transgender women and providers alike report an anecdotal improved breast and/or areolar development, mood, or libido with the use of progestogens.[17,18] There is no evidence to suggest that using progestogens in the setting of transgender care are harmful. In reality some patients may respond favorably to progestogens while others may find negative effects on mood. While progestogens have some anti-androgen effect through central blockade of gonadotropins, there is also a theoretical risk of a direct androgenizing effect of progestogens. This class includes micronized bioidentical progesterone (Prometrium) as well as a number of synthetic progestins. The most commonly used synthetic progestin in the context of transgender care is the oral medroxyprogesterone acetate (Provera).
While concerns exist from the Women's Health Initiative (WHI) regarding risks of cardiovascular disease and breast cancer in the setting of medroxyprogesterone use, these concerns likely do not apply in the context of transgender care for several reasons. First, the transgender women may be at lower risk of breast cancer than non-transgender women. Second, this arm of the WHI involved the use of conjugated equine estrogens in combination with medroxyprogesterone in a sample of menopausal women, some of whom were as long as 10 years post-menopausal at the time of hormone initiation. Third, while statistically significant, the clinical significance of the findings in the WHI was subtle at best. The study aimed to evaluate the role of menopausal hormone therapy in the prevention of chronic disease. The actual findings in the conjugated equine estrogen plus medroxyprogesterone group were an excess absolute risk per 10 000 person-years of 7 more cardiac events, 8 more strokes, 8 more pulmonary emboli, and 8 more invasive breast cancers, with no change in overall mortality.[19] As such this arm of the WHI was stopped early, and it was concluded that combined menopausal hormone therapy is not indicated for prevention of chronic disease.
In the setting of gender affirming care, there are numerous differences to the findings of the WHI: populations tend to be younger, equine estrogens are not used, and the emphasis is on gender affirming interventions which have numerous benefits on mental health and quality of life, rather than prevention. Considering these differences in demographics and goals of therapy, extremely modest increase in overall risk, and lack of difference in mortality, as well as more recent reassuring data with other forms of estrogen, the risks of using progestogens in transgender women are likely minimal or even absent (Grading: NT O M). Injected depo-medroxyprogesterone acetate (Depo-Provera®) is less commonly used in transgender women. Other synthetic progestins may be used as necessitated by formulary limitations; some evidence suggests that norpregnane-derived progestins (norethindrone, norgestrel) may have an increased risk of venous thromboembolism.[20]
| Hormone | Initial-lowb | Initial | Maximumc | Comments |
|---|---|---|---|---|
| ||||
| Estradiol oral | 1mg/day | 2-4mg/day | 8mg/day | if >2mg recommend divided bid dosing |
| Estradiol transdermal | 50mcg | 100mcg | 100-400 mcg | Max single patch dose available is 100mcg. Frequency of change is brand/product dependent. More than 2 patches at a time may be cumbersome for patients |
| Estradiol valerate IM/SQa | 2mg q wk | 4mg q wk | 20mg q wk | |
| Estradiol cypionate IM/SQ | 1mg q wk | 2mg q wk | 5mg q wk | |
| Hormone | Initial-lowb | Initial | Maximumc |
|---|---|---|---|
| |||
| Medroxyprogesterone acetate (Provera) | 2.5mg qhs | 5-10mg qhs | |
| Micronized progesterone | 100-200mg qhs | ||
| Hormone | Initial-lowb | Initial | Maximumc |
|---|---|---|---|
| |||
| Spironolactone | 25mg qd | 50mg bid | 200mg bid |
| Finasteride | 1mg qd | 5mg qd | |
| Dutasteride | 0.5mg qd | ||
| Test | Comments | Baseline | 3 months* | 6 months* | 12 months* | Yearly | |
|---|---|---|---|---|---|---|---|
| * In first year of therapy only ** Used to calculate bioavailable testosterone; monitoring bioavailable testosterone is optional and may be helpful in complex cases (see text) | |||||||
| BUN/Cr/K+ | Only if spiro used | X | X | X | X | X | |
| Lipids | No evidence to support monitoring at any time; use clinician discretion | Based on USPSTF guidelines | |||||
| A1c or glucose | No evidence to support monitoring at any time; use clinician discretion | Based on USPSTF guidelines | |||||
| Estradiol | X | X | X | ||||
| Total Testosterone | X | X | X | X | |||
| Sex Hormone Binding Globulin (SHBG)** | PRN | ||||||
| Albumin** | PRN | ||||||
| Prolactin | Only if symptoms of prolactinoma | ||||||
Overview of titration and monitoring
The interpretation of hormone levels for transgender individuals is not yet evidence based; physiologic hormone levels in non-transgender people are used as reference ranges. However, estrogen levels in non-transgender women may not be associated with specific secondary sex characteristics (i.e. higher estrogen levels in non-transgender women are not necessarily associated with larger breasts), and specific phenotypical end points are likely multifactorial and particularly dependent on genetics and the age at which gender affirming hormone therapy is begun. Titration upwards of dose should be driven by patient goals, in the context of clinical response, hormone level monitoring, and safety monitoring (e.g. presence of risk factors such as smoking, renal function and K+ in patients using spironolactone). A general approach for titration would include increasing of both estrogen and antiandrogen dosing until the estrogen dose is in the female physiologic range. Once this has been achieved, titration efforts can focus on increasing androgen blockade. There can be several approaches to titration of androgens. One approach is to continue increasing estrogen until it reaches the upper limit of the female physiologic range. The drawback for this approach is that patients may begin to experience estrogenic side effects as described below. Another approach is to maintain current physiologic estrogen dosing and titrate upward on antiandrogens and/or addition of a progestogen.
Some providers choose to omit the use of hormone level testing and only monitor for clinical progress or changes. The risk of this approach is that if hormone levels (particularly testosterone) have not reached the target range, but progress is judged as appropriate based on clinical exam, a suboptimal degree of feminization is possible, and the presence of supraphysiologic levels would also be obscured. Conversely, Endocrine Society guidelines recommend monitoring of hormone levels every 3 months.[21] In practice this is not realistic and not likely to add value once a stable dosing has been achieved. A prospective study of transgender women taking 4mg/day divided dose oral estradiol or 100mcg transdermal estradiol, plus 100-200mg/day divided dose spironolactone found that all women achieved physiologic estradiol levels, though only 2/3 of the women achieved female range testosterone levels.[22] Some gender-nonconforming/nonbinary patients may prefer to maintain estradiol or testosterone levels in an intermediate range. Regardless of initial dosing scheme chosen, dosing may be titrated upwards over 3-6 months. Check estradiol and testosterone levels at 3 and 6 months and titrate dose accordingly. For those patients using spironolactone, check renal function and K+ at 3 months and 6 months, then q 6-12 months. While laboratory monitoring of hormone levels may seem complex, it is of similar difficulty to the monitoring of other similarly complex lab-monitored conditions managed by primary care providers, such as thyroid disorders, anticoagulation, or diabetes.
Once hormone levels have reached the target range for a specific patient, it is reasonable to monitor levels yearly, or only as needed as described below. As with other situations involving maintenance of hormone therapy (menopause, contraception), annual visits are sufficient for transgender women on a stable hormone regimen. Other reasons for measuring hormone levels in the maintenance phase include significant metabolic shifts such as the onset of diabetes or a thyroid disorder, substantial weight changes, subjective or objective evidence of virilization, or new symptoms potentially precipitated or exacerbated by hormone imbalances such as hot flashes or migraines. Such patients may also require more frequent office visits to manage coexisting conditions. Increased frequency of office visits may also be useful for patients with complex psychosocial situations to allow for the provision of ancillary or wraparound services.
Current Endocrine Society recommendations include the measurement of only total testosterone and estradiol. This is consistent with Endocrine Society recommendations that only total testosterone be monitored in non-transgender men being managed for testosterone deficiency, except in cases of borderline testosterone levels. However, since testosterone is of particular concern is insuring maximal feminization, the calculation of bioavailable testosterone in transgender women may still be of value. Specifically, exogenous estrogens (especially oral) may be associated with elevated levels of sex hormone binding globulin (SHBG); such elevations can vary from person to person and across regimens. As such in cases of patient concern or persistent virilized features in the presence of a female-range total testosterone, calculation of the bioavailable testosterone may help fine tune hormone regimens for optimal effect.
Interpretation of laboratory results requires special attention in the context of transgender care. Numerous sources publish target ranges for serum estradiol, total estrogens, free, total and bioidentical testosterone, and sex hormone binding globulin. However, these specific ranges may vary between different laboratories and techniques. Furthermore, the interpretation of reference ranges supplied with lab result reports may not be applicable if the patient is registered under a gender that differs from their intended hormonal sex. For example, a transgender woman who is still registered as male will result in lab reference ranges reported for a male; clearly these ranges are not applicable for a transgender woman using feminizing hormone therapy. Hormone levels for genderqueer or gender nonconforming/nonbinary patients may intentionally lie in the mid-range between male and female norms. Providers are encouraged to consult with their local lab(s) to obtain hormone level reference ranges for both ‘male’ and ‘female’ norms, and then apply the correct range when interpreting results based on the current hormonal sex, rather than the sex of registration.
Monitoring estradiol levels
Historically estrogen levels have been monitored using the total serum estradiol. The 2009 Endocrine Society Guidelines recommend monitoring serum estradiol and maintaining levels at the mid-cycle range for non-transgender women.[21] This recommendation is based on expert opinion only and may be overly conservative, and hormone levels are often not easy to tightly control.[23] Providers are encouraged to review the specific estradiol reference ranges for their local lab estradiol assays, as these can vary. There is no evidence that higher estradiol levels in patients with adequate androgen suppression results in additional feminization or breast development. Maintaining estrogen levels in the physiologic range for menstruating non-transgender women minimizes risks and side effects, and makes sense clinically. Note that the use of conjugated estrogens (Premarin®) or ethinyl estradiol (found in most combined oral contraceptives) are not accurately measured by estradiol assays and will typically result in low measured levels.
In patients who have been using self-administered conjugated estrogens, or ethinyl estradiol, it is reasonable to check a total estrogens level, which may provide a more accurate estimate in these cases. This assay also measures minor estrogens such as estriol and estrone. There is some evidence that the use of oral estradiol results in higher serum levels of estrone due to first pass hepatic metabolism, as compared to parenteral forms.[24,25] This may explain dose independent reasons why some patients "feel different" on different forms of estrogen.
Monitoring testosterone levels
Testosterone levels can be difficult to measure in non-transgender men due to rapid fluctuations in levels, relating to pulsatile release of gonadotropins, with higher levels in the morning hours. Free testosterone represents the portion of testosterone unbound to serum proteins and depends on levels of sex hormone binding globulin (SHBG). While free testosterone can be measured, assays are unreliable.[26] Consensus is lacking on the role of free vs. total testosterone levels; total testosterone levels are reliable and readily available, however they do not describe the actual bioavailable testosterone level. Bioavailable testosterone is free testosterone plus testosterone weakly bound to albumin.[27] SHBG is elevated in the presence of estrogen, and in particular with exogenous estrogen supplementation, more so with oral estrogen than with parenteral routes due to first pass hepatic activity.[28] For transgender care, The Endocrine Society recommends monitoring of the total testosterone level, with a target range of <55ng/dl .[21] Calculation of the bioavailable testosterone may help guide dosing, and can be calculated from the total testosterone, albumin, and SHBG levels.[29] A general reference range for bioavailable testosterone is >72ng/dl (2.5nmol/L).[30-32]
Monitoring gonadotropin levels
When indicated, measuring of gonadotropins (luteinizing hormone: LH and follicle stimulating hormone: FSH) can be done using the local lab ranges for eugonadal state as a reference.
Monitoring hormone levels in patients using injected estrogen
Pharmacokinetic studies of injected estrogen have been limited. Two earlier studies only examined single-dose pharmacokinetics and are therefore unable to be applied to steady-state dosing.[33] Studies of estradiol levels in the context of a monthly combined injectable contraceptive of 5mg estradiol cypionate and 25mg medroxyprogesterone acetate found peak levels 2-4 days after injection, maximum estradiol levels of approximately 250 pg/ml, and trough levels of approximately 50pg/ml.[34,35] These findings suggest that injected estradiol in the middle of the dosing ranges recommended here will result in physiologic estradiol levels, and that use of more frequent dosing will reduce peak-trough effect. When measuring hormone levels in patients using injected forms of estradiol, a mid-cycle level is often sufficient, however if the patient is experiencing cyclic symptoms such as migraines or mood swings, peak (1-2 days post injection) and trough levels of both estradiol and testosterone may reveal wide fluctuations in hormone levels over the dosing cycle; in these cases, consider changing to an oral or transdermal preparation, or reducing the injection interval (with concomitant reduction in dose, to maintain the same total dose administered over time). A single study suggests similar pharmacokinetics when estradiol is injected subcutaneously, rather than intramuscular.[34]
Interpreting sex-specific, non-hormone labs
Alkaline phosphatase, hemoglobin and hematocrit (H&H), and creatinine may vary depending on the patient's current sex hormone configuration. Several factors contribute to these differences, bone mass, muscle mass, number of myocytes, presence or lack of menstruation, and the erythropoietic effect of testosterone. While transgender women do not menstruate, those with female-range hormone levels will lack the erythropoietic effects of male-range testosterone, and it may be reasonable to use the female-range lower limit of normal when interpreting H&H. Conversely, the lack of menstruation, and potential for pulsatile undetected androgen activity in those with retained gonads make it reasonable to use the male-range upper limit of normal for H&H. Using the male-range upper limit of normal for alkaline phosphatase and creatinine may also be appropriate for transgender women due to retained bone and muscle mass or myocyte counts, respectively. This is of particular importance in transgender women using spironolactone who are registered as female, and may have a lab result flag showing an abnormal elevated creatinine. In these cases the provider should reference the male normal ranges for their lab.[19]
| Lab measure | Lower Limit of normal | Upper Limit of normal |
|---|---|---|
| Creatinine | Not defined | Male value |
| Hemoglobin/Hematocrit | Female value | Male value |
| Alkaline Phosphatase | Not defined | Male value |
Individualized dosing based on patient centered goals
Some patients may desire limited hormone effects or a mix of masculine and feminine sex characteristics. Examples include retention of erectile function with otherwise maximum feminization, or minimal feminizing effects with the exception of body or facial hair elimination or breast growth. While manipulation of dosing regimens and choice of medication can allow patients to achieve this goal, it is important to have a clear discussion with patients regarding expectations and unknowns. Specifically, it is not possible to select in advance an exact hormone regimen that will predictably allow patients to arrive at a specified configuration of sex characteristics. Furthermore, individual genetic and physiologic variation can result in wide variations in both blood levels and response to therapy between different individuals using the same route and dose. The best approach in these cases is to start with low doses and advance slowly, titrating to effect. At the same time, response to hormone therapy is also individualized and measures such as breast growth are variable in both degree and time course. Likely predictive factors of speed and degree of feminization include genetics, age at initiation of therapy, and body habitus.[17] Patients should be counseled on typical timeframes for changes and advised to avoid making comparisons to the experiences of others. Anecdotal sources suggest that maximal feminization may occur within 2-5 years.[36]
Specific considerations and conditions
Tobacco use:
Tobacco use in combination with estrogen therapy is associated with an increased risk of venous thromboembolism. All transgender women who smoke should be counselled on tobacco risks and cessation options at every visit. Many transgender women may be unable or unwilling to quit smoking; this should not represent an absolute contraindication to estrogen therapy. After an in depth and careful informed consent discussion, it is reasonable to prescribe estrogen using a harm reduction approach, with a preferred route of transdermal estrogen. Aspirin 81mg/day can be considered as an additional preventive measure in smokers, though no evidence exists to allow and informed assessment of the risk/benefit ratio between VTE prevention and gastrointestinal hemorrhage (Grading: X C W). Transdermal estrogens are preferred to minimize risk (Grading: T O S).
Loss of erectile function:
Sildenafil (Viagra) and tadalafil (Cialis) can be used for preservation of erectile function at any stage or with any feminizing hormone regimen, in consideration of the typical contraindications and precautions when using this class of medication. Individual results may vary. It is reasonable check both total and bioavailable testosterone levels, and consider reduction of androgen blockade to allow an increase in testosterone, depending on patient goals.
Low libido:
A study of sexual desire in transgender women found that 83% never or rarely experience spontaneous sexual desire, 76% never or rarely experience responsive sexual desire, and 22% meet the criteria for Hypoactive Sexual Desire Disorder (HSDD) by experiencing both of these in a way which results in personal or relational distress. This study also found decreases in sexual desire after genital surgery.[37] Another study found a rate of HSDD in transgender women of 34%, compared to 23% in non-transgender women. This study found no correlation between sexual desire and testosterone levels in the transgender women, though a significant correlation was found between hormones and desire in non-transgender women.[38] An unpublished study found positive correlations between libido and testosterone levels in transgender women treated with testosterone, but no effect when treated with dehydroepiandrosterone sulfate (DHEA-S).[39] As such it remains unclear if HSDD relates to androgen blockade or post-gonadectomy hormonal changes, or due to anatomical, functional and psychological changes associated with hormone therapy or genital surgery.
Post-gonadectomy:
Since estrogen dosing should be based on physiologic female levels, no reduction in estrogen dosing is required after gonadectomy. Some patients may choose to use a lower dose, which is appropriate as long as dosing is adequate to maintain bone density. Adequacy of dosing in those on low estrogen replacement post gonadectomy may be assessed by following LH and FSH levels (Grading: T O W).[11]
Older transgender women:
Older transgender women initiating therapy may have less rapid and a lesser degree of changes. Due to higher levels of co-occurring conditions in older individuals, there may also be higher risk of adverse effects. Nevertheless a large number of women have started hormones at advanced ages and safety and satisfaction have been reported as acceptable.[40] There is no evidence to support continuation or cessation of hormones for older transgender women.
Since the mean age of menopause in the U.S. is 49,[41] it is reasonable in transgender women who have undergone gonadectomy to consider stopping hormone therapy around age 50. Expected effects of this may be similar to non-transgender women experiencing menopause. Transgender women who retain their gonads but withdraw hormone therapy may experience return of virilization. A discussion of the pros and cons of this approach, with individualized and shared decision making is recommended.
Pituitary adenoma (prolactinoma) and galactorrhea:
Prolactin elevations and growth of pituitary prolactinomas are theoretical risks associated with estrogen therapy; several cases have been reported.[42] However, with the administration of physiologic doses of estrogen, there is no clear basis for an increased risk of prolactinomas in comparison to the population background rate in non-transgender women. Furthermore, Endocrine Society guidelines for the management of incidental prolactinomas are expectant management only, in the absence of suggestive visual or other symptoms (significant galactorrhea, headaches).[43] Routine screening with serum prolactin levels in asymptomatic transgender women would not have an impact on management, and could result in costs or harm if further workup if pursued. As such it is recommended that prolactin be checked only in cases of visual disturbances, excessive galactorrhea, and be considered in cases of new onset headaches. It is noted that some transgender women experience a minimal amount of galactorrhea early in their hormone therapy course. The presence of non-bloody minimal galactorrhea from more than one duct and/or bilateral is almost certainly physiologic and would not warrant further evaluation.
Venous thromboembolism:
Data from studies of menopausal women suggest no increased risk of venous thromboembolism with the use of transdermal estradiol.[44] There are some data suggestive of increased thrombogenicity and cardiovascular risk when conjugated equine estrogens (Premarin) are used.[1,2] Data on the risk associated with oral 17-beta estradiol are mixed, with some suggesting no increased risk and others suggesting a 2.5 - 4 fold increased risk.[20,44] Even in the case of a 2.5 fold increase, the background rate for VTE in the general population is very low (1 in 1000 to 1 in 10,000), so the absolute risk increase is minimal.[3] There is weak evidence that sublingual administration of oral estradiol tablets might reduce thromboembolic risk due to a bypass of hepatic first pass, with one study showing 13 fold increase in peak estradiol blood levels but similar 24 hour area-under-the-curve.[45] A study of sublingual estradiol for the management of post-partum depression found that it was well tolerated, and the increased pulsatile nature of this route may more closely mimics natural ovarian estrogen secretion.[46] Sublingual administration requires insuring that the estradiol tablets are micronized; while most commonly available estradiol tablets are micronized, specifying as such on the written prescription (or consulting with the dispensing pharmacist) is recommended. Conversely, the overall risk of taking oral estradiol is low, and patients using sublingual estradiol may experience wide swings in hormone levels, inconsistent absorption, and more difficulty suppressing testosterone via feedback inhibition.
There is also some limited evidence to suggest that the risk of VTE in menopausal women may be driven more by the choice of progestogen, and that pregnane derived progestogens such as medroxyprogesterone in combination with oral estradiol does not confer an increased risk, while norpregnane derived progestogens such as norethindrone may increase risk by 80% when used with oral estradiol.[20] Prior studies reporting a 20 to 40 fold increased risk of VTE in transgender women involved the use of high doses (100-200mcg/day) of thrombogenic ethinyl estradiol in a mix of smokers and non-smokers.[47,48] A retrospective cohort of Dutch transgender women found no increased risk in VTE once ethinyl estradiol was replaced by bioidentical estradiol as the standard regimen.[49]
Insufficient evidence exists to definitively guide estrogen therapy in transgender women with risk factors or with a personal history of prior VTE, either on or off estrogen. A report of 11 transgender women with a history of activated protein C resistance (the mechanism of action implicated in the hypercoagulable state associated with the Factor-V Lieden mutation) using transdermal estradiol without anticoagulation found no clotting events after a mean of 64 months of therapy.[50]
Figures 1-5 describe the approach to various scenarios of VTE history or risk factors and estrogen use. The decision to initiate episodic (i.e. before long airplane flights) or long term anticoagulation or antiplatelet therapy should be considered in the context of risks associated with major gastrointestinal or intracranial hemorrhage. Routine VTE prophylaxis with aspirin in unselected transgender populations is not recommended. Routine screening for prothrombotic mutations is not recommended in the absence of risk factors.[50] Regardless of the circumstances, estrogen therapy should not be administered in patients with significant risk factors for or history of VTE who continue to smoke tobacco.
Figure 1. Approach to management of estrogen in patients with a personal history of VTE

This figure outlines the estrogen management approaches for patients with a personal history of VTE.
-
Patients with positive prothrombotic mutations or other evidence of hypercoagulative state should begin anticoagulation as per current guidelines for nontransgender patients. Begin transdermal estrogen after informed consent discussion.
-
For patients without positive prothrombotic mutations or evidence of hypercoagulative state, determine if there is a clear external cause for VTE such as long bone fracture, immobility, or tobacco use. If yes, continue with standard approach to estrogen therapy once external cause has been resolved. If no, consider episodic or long-term anticoagulation or antiplatelet therapy on a case-by-case basis, using the same criteria as for nontransgender patient.
Figure 2. Approach to management of estrogen in patients with a family history of VTE but no personal history of VTE

Approach to management of estrogen in patients with a family history of VTE but no personal history of VTE. Treat as nontransgender patients. Consider treatment per guidelines.
Figure 3. Approach to patient using oral or injected estrogen at time of first diagnosis of VTE

Discusses decision points related to determining cause of VTE and use of anticoagulation agents before beginning transdermal estrogen therapy.
Figure 4. Approach to patient using transdermal estradiol at time of first diagnosis of VTE
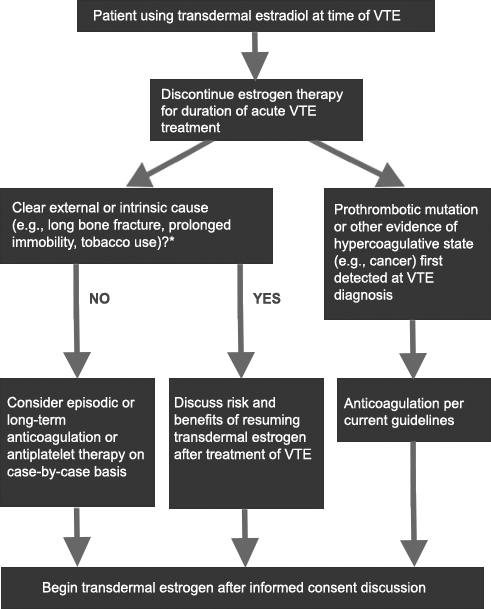
Discusses decision points for treatment of patients taking transdermal estradiol at time of first diagnosis of VTE and whether to consider longterm anticoagulation or anti-platelet therapy.
Figure 5. Approach to patients with known hypercoagulative state who use transdermal estradiol and present with acute VTE

Decision tree for patients with known hypercoagulative state who present with acute VTE and use transdermal estradiol. Those already anitcoagulated should consider lifelong avoidance of estrogen therapy. Others should begin long-term anticoagulation therapy unless contraindicated.
Autoimmunity:
There is a certain but incompletely defined linkage between sex hormones and autoimmune conditions. Testosterone has been associated with overall immune suppression, and autoimmune conditions are more common in non-transgender women than men.[51] Testosterone deprivation results in an increased Th1:Th2 ratio.[52] However the relationship is more complex, as demonstrated by the paradoxical improvements seen in multiple sclerosis during pregnancy.[51] In transgender women who have undergone orchiectomy or have full androgen blockade, some evidence suggests that supplementation with dehydroepiandrosterone (DHEA) may counteract some of the shift toward autoimmunity.[53] Patients with autoimmune conditions should be informed that their condition could potentially worsen (or improve) once feminizing therapy has begun. Hormone dosing should begin low and advance slowly, monitoring for worsening symptoms, and in collaboration with any specialists who may be managing the autoimmune condition.
Migraine:
Migraines have a clear hormonal component and may be exacerbated by estrogen therapy. Patients with a history of migraines should consider starting with a low dose and titrating upward as tolerated. Oral or transdermal estrogen may be preferred to the potentially cyclic levels associated with injected estrogen.[54] While migraine with aura is associated with an increased risk of stroke in women using oral contraceptives,[55] it is not clear if this risk translates to the use of bioidentical estradiol.
Mental health conditions:
While hormones may contribute to mood disorders (such as in premenstrual dysphoric disorder or postpartum depression), there is no clear evidence that estrogen therapy is directly associated with the onset of or worsening of mental health conditions. In fact one study found that transgender women experience improvements in social functioning and reduced anxiety and depression once estrogen therapy is begun.[56] Mental health conditions in transgender women should be approached with a broad differential diagnosis as in any other patient. It may be advisable to avoid injected estrogen due to the potentially cyclic levels, which could bring about or worsen existing mood symptoms.
Estrogen therapy in patients with a prior history of cancer:
An active estrogen-sensitive cancer is a contraindication to estrogen therapy. For patients with a prior history of estrogen sensitive cancer (breast, pituitary), consultation with an oncologist is recommended. While androgen deprivation is a mainstay of treatment for advanced prostate cancer, it is unclear if estrogen therapy may confer an independent protection or increased risk of prostate cancer.[57] PSA should be considered unreliable in those using antiandrogen or estrogen therapy due to the high risk of false negative tests.
Perioperative use of feminizing hormones:
No direct study of the risk of perioperative venous thromboembolism in users of bioidentical estrogens has been conducted. Guidelines from two British professional organizations make a weak recommendation to discontinue menopausal hormone therapy in the perioperative period, however both acknowledge that this may not be needed in the setting of proper prophylaxis (i.e. heparin or compression devices).[58] Studies of perioperative ethinyl estradiol in users of hormonal contraception have mixed findings and are wrought with confounding and methodological limitations.[59] Many surgeons insist that transgender women discontinue estrogen for several weeks before and after any gender affirming procedure.[60,61] These recommendations may appear as benign to the surgeon; however to the transgender woman undergoing a life and body-altering procedure simultaneous with gonadectomy, sudden and prolonged complete withdrawal of estrogens can have a profound impact. Postoperative depression is a nontrivial concern and may have some basis in the drastic hormone shifts, including cessation of estrogens, experienced in the perioperative period. There is no evidence to suggest that transgender women who lack specific risk factors (smoking, personal or family history, excessive doses or use of synthetic estrogens) must cease estrogen therapy before and after surgical procedures, in particular with appropriate use of prophylaxis and an informed consent discussion of the pros and cons of discontinuing hormone therapy during this time. Possible alternatives include using a lower dose of estrogen, and/or changing to a transdermal route if not already in use.[62]
About consent forms for hormone therapy:
Informed consent is a process which occurs between a patient and a provider. The process should include an individualized discussion of the risks, benefits, unknowns, alternatives, and risk of no treatment. We are no longer recommending the use of consent forms for hormone therapy. Many other common interventions such as contraception or HIV pre-exposure prophylaxis do not involve the use of consent forms, and rely on a discussion and shared decision making between patient and provider. If the informed consent process is properly documented in the chart, consent forms do not likely provide any additional legal protections to the provider. Elimination of consent forms helps to demystify and destigmatize hormone therapy. Providers can use the information provided in these guidelines to frame their individualized discussions with patients.
Hormone therapy information for patients
References
- Shifren JL, Rifai N, Desindes S, McIlwain M, Doros G, Mazer NA. A comparison of the short-term effects of oral conjugated equine estrogens versus transdermal estradiol on C-reactive protein, other serum markers of inflammation, and other hepatic proteins in naturally menopausal women. J Clin Endocrinol Metab. 2008 May;93(5):1702-10.
- Ho JY-P, Chen M-J, Sheu WH-H, Yi Y-C, Tsai AC-W, Guu H-F, et al. Differential effects of oral conjugated equine estrogen and transdermal estrogen on atherosclerotic vascular disease risk markers and endothelial function in healthy postmenopausal women. Hum Reprod Oxf Engl. 2006 Oct;21(10):2715-20.
- Hugon-Rodin J, Gompel A, Plu-Bureau G. Mechanisms in endocrinology: epidemiology of hormonal contraceptives-related venous thromboembolism. Eur J Endocrinol. 2014 Dec 1;171(6):R221-30.
- Asscheman H, Giltay EJ, Megens JAJ, de Ronde WP, van Trotsenburg MAA, Gooren LJG. A long-term follow-up study of mortality in transsexuals receiving treatment with cross-sex hormones. Eur J Endocrinol Eur Fed Endocr Soc. 2011 Apr;164(4):635-42.
- Koetsawang S, Mandlekar AV, Krishna UR, Purandare VN, Deshpande CK, Chew SC, et al. A randomized, double-blind study of two combined oral contraceptives containing the same progestogen, but different estrogens. Contraception. 1980 May;21(5):445-59.
- Prior JC, Vigna YM, Watson D. Spironolactone with physiological female steroids for presurgical therapy of male-to-female transsexualism. Arch Sex Behav. 1989 Feb;18(1):49-57.
- Sinclair R, Patel M, Dawson TL, Yazdabadi A, Yip L, Perez A, et al. Hair loss in women: medical and cosmetic approaches to increase scalp hair fullness. Br J Dermatol. 2011 Dec;165 Suppl 3:12-8.
- Rittmaster RS. 5alpha-reductase inhibitors. J Androl. 1997 Dec;18(6):582-7.
- Traish AM, Hassani J, Guay AT, Zitzmann M, Hansen ML. Adverse side effects of 5α-reductase inhibitors therapy: persistent diminished libido and erectile dysfunction and depression in a subset of patients. J Sex Med. 2011 Mar;8(3):872-84.
- Bienz M, Saad F. Androgen-deprivation therapy and bone loss in prostate cancer patients: a clinical review. BoneKEy Rep. 2015;4:716.
- Esteva I, Yahyaoui R, Cano G, Giraldo F, Bergero T, Ruiz de Adana S, et al. Evolution of gonadal axis after sex reassignment surgery in transsexual patients in the Spanish public health system. Int J Transgenderism. 2006 Jun 1;9(2):15-22.
- Bessone F, Lucena MI, Roma MG, Stephens C, Medina-Cáliz I, Frider B, et al. Cyproterone acetate induces a wide spectrum of acute liver damage including corticosteroid-responsive hepatitis: report of 22 cases. Liver Int Off J Int Assoc Study Liver. 2016 Feb;36(2):302-10.
- Kolvenbag GJCM, Blackledge GRP. Worldwide activity and safety of bicalutamide: a summary review. Urology. 1996 Jan;47(1, Supplement 1):70-9.
- Wolf-Gould CS, Wolf-Gould CH. A transgender woman with testicular cancer: a new twist on an old problem. LGBT Health. 2015 Dec 24;
- Rosenthal SM. Approach to the patient: transgender youth: endocrine considerations. J Clin Endocrinol Metab. 2014 Dec;99(12):4379-89.
- Dittrich R, Binder H, Cupisti S, Hoffmann I, Beckmann MW, Mueller A. Endocrine treatment of male-to-female transsexuals using gonadotropin-releasing hormone agonist. Exp Clin Endocrinol Diabetes Off J Ger Soc Endocrinol Ger Diabetes Assoc. 2005 Dec;113(10):586-92.
- Wierckx K, Gooren L, T'Sjoen G. Clinical review: Breast development in trans women receiving cross-sex hormones. J Sex Med. 2014 May;11(5):1240-7.
- Orentreich N, Durr NP. Proceedings: Mammogenesis in transsexuals. J Invest Dermatol. 1974 Jul;63(1):142-6.
- Writing Group for the Women's Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the women's health initiative randomized controlled trial. JAMA. 2002 Jul 17;288(3):321-33.
- Canonico M, Oger E, Plu-Bureau G, Conard J, Meyer G, Levesque H, et al. Hormone therapy and venous thromboembolism among postmenopausal women: impact of the route of estrogen administration and progestogens: the ESTHER Study. Circulation. 2007 Feb;115(7):840-5.
- Hembree WC, Cohen-Kettenis P, Delemarre-van de Waal HA, Gooren LJ, Meyer WJ, Spack NP, et al. Endocrine treatment of transsexual persons: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2009 Jun 9;94(9):3132-54.
- Deutsch MB, Bhakri V, Kubicek K. Effects of cross-sex hormone treatment on transgender women and men. Obstet Gynecol. 2015 Mar;125(3):605-10.
- Quest Diagnostics. Estradiol. [cited 2015 Nov 20]. Test Center Info - For Physicians & Hospitals
- Ansbacher R. The pharmacokinetics and efficacy of different estrogens are not equivalent. Am J Obstet Gynecol. 2001 Feb;184(3):255-63.
- O'Connell MB. Pharmacokinetic and pharmacologic variation between different estrogen products. J Clin Pharmacol. 1995 Sep;35(9 Suppl):18S - 24S.
- Ly LP, Handelsman DJ. Empirical estimation of free testosterone from testosterone and sex hormone-binding globulin immunoassays. Eur J Endocrinol Eur Fed Endocr Soc. 2005 Mar;152(3):471-8.
- Rosner W, Auchus RJ, Azziz R, Sluss PM, Raff H. Position statement: Utility, limitations, and pitfalls in measuring testosterone: an Endocrine Society position statement. J Clin Endocrinol Metab. 2007 Feb;92(2):405-13.
- Serin IS, Ozçelik B, BaŞbuğ M, Aygen E, Kula M, Erez R. Long-term effects of continuous oral and transdermal estrogen replacement therapy on sex hormone binding globulin and free testosterone levels. Eur J Obstet Gynecol Reprod Biol. 2001 Dec 1;99(2):222-5.
- Carnegie C. Diagnosis of hypogonadism: clinical assessments and laboratory tests. Rev Urol. 2004;6(Suppl 6):S3-8.
- Sih R, Morley JE, Kaiser FE, Perry HM, Patrick P, Ross C. Testosterone replacement in older hypogonadal men: a 12-month randomized controlled trial. J Clin Endocrinol Metab. 1997 Jun;82(6):1661-7.
- Korenman SG, Morley JE, Mooradian AD, Davis SS, Kaiser FE, Silver AJ, et al. Secondary hypogonadism in older men: its relation to impotence. J Clin Endocrinol Metab. 1990 Oct;71(4):963-9.
- Nelson R, O'Kane D, Heser D, Klee G. A simple and rapid assay for bioavailable-testosterone. Clin Chem. 2001 Jun;47(6):A20-A20.
- Düsterberg B, Nishino Y. Pharmacokinetic and pharmacological features of oestradiol valerate. Maturitas. 1982 Dec;4(4):315-24.
- Sierra-Ramírez JA, Lara-Ricalde R, Lujan M, Velázquez-Ramírez N, Godínez-Victoria M, Hernádez-Munguía IA, et al. Comparative pharmacokinetics and pharmacodynamics after subcutaneous and intramuscular administration of medroxyprogesterone acetate (25 mg) and estradiol cypionate (5 mg). Contraception. 2011 Dec;84(6):565-70.
- Thurman A, Kimble T, Hall P, Schwartz JL, Archer DF. Medroxyprogesterone acetate and estradiol cypionate injectable suspension (Cyclofem) monthly contraceptive injection: steady-state pharmacokinetics. Contraception. 2013 Jun;87(6):738-43.
- Coleman E, Bockting W, Botzer M, Cohen-Kettenis P, DeCuypere G, Feldman J, et al. Standards of Care for the Health of Transsexual, Transgender, and Gender-Nonconforming People, Version 7. Int J Transgenderism. 2012;13(4):165-232.
- Wierckx K, Elaut E, Van Hoorde B, Heylens G, De Cuypere G, Monstrey S, et al. Sexual desire in trans persons: associations with sex reassignment treatment. J Sex Med. 2014 Jan;11(1):107-18.
- Elaut E, De Cuypere G, De Sutter P, Gijs L, Van Trotsenburg M, Heylens G, et al. Hypoactive sexual desire in transsexual women: prevalence and association with testosterone levels. Eur J Endocrinol Eur Fed Endocr Soc. 2008 Mar;158(3):393-9.
- Sivakumaran D, Seal L. A retrospective study on the use of androgen therapy in trans women to treat HSSD. EPATH conference; 2015 Mar 13; Ghent, Belgium.
- Gooren L, Lips P. Conjectures concerning cross-sex hormone treatment of aging transsexual persons. J Sex Med. 2014 Aug;11(8):2012-9.
- Tom SE, Cooper R, Patel KV, Guralnik JM. Menopausal Characteristics and Physical Functioning in Older Adulthood in the NHANES III. Menopause N Y N. 2012 Mar;19(3):283-9.
- Goh HH, Li XF, Ratnam SS. Effects of cross-gender steroid hormone treatment on prolactin concentrations in humans. Gynecol Endocrinol Off J Int Soc Gynecol Endocrinol. 1992 Jun;6(2):113-7.
- Freda PU, Beckers AM, Katznelson L, Molitch ME, Montori VM, Post KD, et al. Pituitary incidentaloma: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2011 Apr;96(4):894-904.
- Canonico M, Plu-Bureau G, Lowe G, Scarabin PY. Hormone replacement therapy and risk of venous thromboembolism in postmenopausal women: systematic review and meta-analysis. Bmj. 2008;336(7655):1227.
- Price TM, Blauer KL, Hansen M, Stanczyk F, Lobo R, Bates GW. Single-dose pharmacokinetics of sublingual versus oral administration of micronized 17 beta-estradiol. Obstet Gynecol. 1997 Mar;89(3):340-5.
- Ahokas A, Kaukoranta J, Wahlbeck K, Aito M. Estrogen deficiency in severe postpartum depression: successful treatment with sublingual physiologic 17beta-estradiol: a preliminary study. J Clin Psychiatry. 2001 May;62(5):332-6.
- Asscheman H, Gooren LJG, Eklund PLE. Mortality and morbidity in transsexual patients with cross-gender hormone treatment. Metabolism. 1989;38(9):869-73.
- van Kesteren PJ, Asscheman H, Megens JA, Gooren LJ. Mortality and morbidity in transsexual subjects treated with cross-sex hormones. Clin Endocrinol (Oxf). 1997 Sep;47(3):337-42.
- Asscheman H, Giltay EJ, Megens JAJ, de Ronde W, van Trotsenburg MAA, Gooren LJG. A long-term follow-up study of mortality in transsexuals receiving treatment with cross-sex hormones. Eur J Endocrinol. 2011 Jan 25;164(4):635-42.
- Ott J, Kaufmann U, Bentz EK, Huber JC, Tempfer CB. Incidence of thrombophilia and venous thrombosis in transsexuals under cross-sex hormone therapy. Fertil Steril. 2010;93(4):1267-72.
- Gold SM, Voskuhl RR. [Internet]. Elsevier; 2009 [cited 2015 Nov 21]. p. 239-51. Estrogen and testosterone therapies in multiple sclerosis. In: Progress in Brain Research
- Trigunaite A, Dimo J, Jørgensen TN. Suppressive effects of androgens on the immune system. Cell Immunol. 2015 Apr;294(2):87-94.
- Labrie F. DHEA, important source of sex steroids in men and even more in women. Prog Brain Res. 2010;182:97-148.
- Chai NC, Peterlin BL, Calhoun AH. Migraine and estrogen. Curr Opin Neurol. 2014 Jun;27(3):315-24.
- Nappi RE, Merki-Feld GS, Terreno E, Pellegrinelli A, Viana M. Hormonal contraception in women with migraine: is progestogen-only contraception a better choice? J Headache Pain. 2013;14:66.
- Gómez-Gil E, Zubiaurre-Elorza L, Esteva I, Guillamon A, Godás T, Cruz Almaraz M, et al. Hormone-treated transsexuals report less social distress, anxiety and depression. Psychoneuroendocrinology. 2012 May;37(5):662-70.
- Misawa A, Inoue S. Estrogen-related receptors in breast cancer and prostate cancer. Front Endocrinol. 2015;6:83.
- Royal College of Obstetricians and Gynaecologists. United Kingdom; 2011 May. Report No.: 19. Available from: Venous Thromboembolism and Hormone Replacement Therapy, 3rd edition
- Whitehead EM, Whitehead MI. The pill, HRT and postoperative thromboembolism: cause for concern? Anaesthesia. 1991 Jul 1;46(7):521-2.
- Johnson RL, Hemington-Gorse SJ, Dhital SK. Do cosmetic surgeons consider estrogen-containing drugs to be of significant risk in the development of thromboembolism? Aesthetic Plast Surg. 2008 Apr 30;32(5):743-7.
- Asscheman H, T'Sjoen G, Lemaire A, Mas M, Meriggiola MC, Mueller A, et al. Venous thrombo-embolism as a complication of cross-sex hormone treatment of male-to-female transsexual subjects: a review. Andrologia. 2014 Sep;46(7):791-5.
- Brighouse D. Hormone replacement therapy (HRT) and anaesthesia. Br J Anaesth. 2001 May 1;86(5):709
